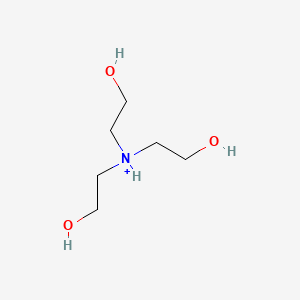
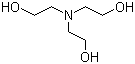
Triethanolamine, (HOCH2CH2)3N, is a widely used biological buffer, with maximum buffering capacity at pH 7.8. Propose a synthesis of this compound from ethylene oxide and ammonia. | Homework.Study.com
SOLVED: Triethanolamine (TEA) buffer Is popular organic buffer. Its pKa Is 7.8 Describe the preparation of a 1-L solution of 0.05 M TEA buffer at PH 7.6. You have availlable at your
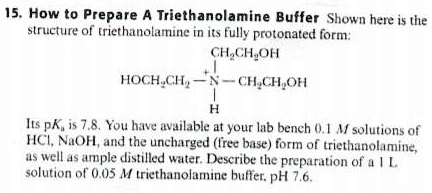
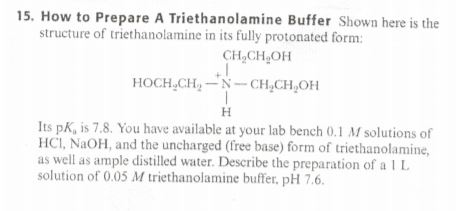
SOLVED: How to Prepare A Triethanolamine Buffer Shown hete is the structure Ol (riethanolamine in Its tully protonated orm; GH,CHLOH HOCH CH, CHCHOH Its pK, is 7. , You have available at