
Mineralogy, nucleation and growth of dolomite in the laboratory and sedimentary environment: A review - Gregg - 2015 - Sedimentology - Wiley Online Library

Low-Temperature Synthesis of Disordered Dolomite and High-Magnesium Calcite in Ethanol–Water Solutions: The Solvation Effect and Implications | ACS Omega

Calcite, dolomite and magnesite dissolution kinetics in aqueous solutions at acid to circumneutral pH, 25 to 150 °C and 1 to 55 atm pCO2: New constraints on CO2 sequestration in sedimentary basins - ScienceDirect

Calcite, dolomite and magnesite dissolution kinetics in aqueous solutions at acid to circumneutral pH, 25 to 150 °C and 1 to 55 atm pCO2: New constraints on CO2 sequestration in sedimentary basins - ScienceDirect

Calcite, dolomite and magnesite dissolution kinetics in aqueous solutions at acid to circumneutral pH, 25 to 150 °C and 1 to 55 atm pCO2: New constraints on CO2 sequestration in sedimentary basins - ScienceDirect

A ) The crystal structure of dolomite and ( B ) dolomite-II. The main... | Download Scientific Diagram
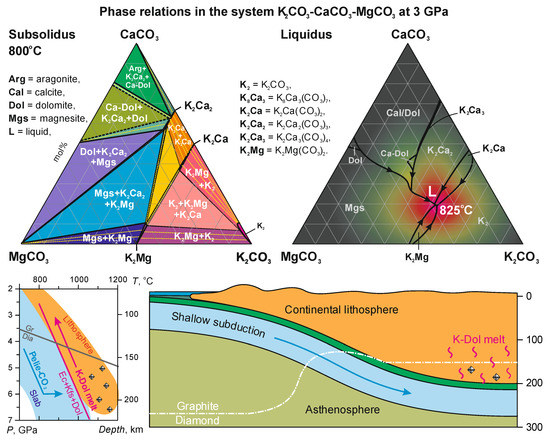
Minerals | Free Full-Text | The System K2CO3–CaCO3–MgCO3 at 3 GPa: Implications for Carbonatite Melt Compositions in the Shallow Continental Lithosphere

Order-disorder-reorder process in thermally treated dolomite samples: A combined powder and single-crystal X-ray diffraction study | Request PDF

Calcite-dolomite solvus representing the temperature at which magnesian... | Download Scientific Diagram

Calcite, dolomite and magnesite dissolution kinetics in aqueous solutions at acid to circumneutral pH, 25 to 150 °C and 1 to 55 atm pCO2: New constraints on CO2 sequestration in sedimentary basins - ScienceDirect

Calcite, dolomite and magnesite dissolution kinetics in aqueous solutions at acid to circumneutral pH, 25 to 150 °C and 1 to 55 atm pCO2: New constraints on CO2 sequestration in sedimentary basins - ScienceDirect

Low-Temperature Synthesis of Disordered Dolomite and High-Magnesium Calcite in Ethanol–Water Solutions: The Solvation Effect and Implications | ACS Omega

New insight into absorption characteristics of CO2 on the surface of calcite, dolomite, and magnesite - ScienceDirect

Calcite, dolomite and magnesite dissolution kinetics in aqueous solutions at acid to circumneutral pH, 25 to 150 °C and 1 to 55 atm pCO2: New constraints on CO2 sequestration in sedimentary basins - ScienceDirect

Calcite–magnesite solid solutions: using genetic algorithms to understand non-ideality | SpringerLink

Metastable structures of CaCO3 and their role in transformation of calcite to aragonite and postaragonite | Crystal Growth & Design
![Location of [aragonite ? magnesite = dolomite] equilibrium determined... | Download Scientific Diagram Location of [aragonite ? magnesite = dolomite] equilibrium determined... | Download Scientific Diagram](https://www.researchgate.net/publication/253651492/figure/fig3/AS:339679305060358@1457997265821/Location-of-aragonite-magnesite-dolomite-equilibrium-determined-in-the-present-in.png)


